Purnima Singh1, Panomootil Blessy Varghese2
Author:
2 Student M.P.T Neurology, Hosmat College of Physiotherapy, RGUHS University, Bangalore, Karnataka, India
Corresponding Author:
1Principal of Hosmat college of Physiotherapy, RGUHS U niversity, Bangalore, Karnataka, India Mail id: purnimasingh29@gmail.com
ABSTRACT
| Introduction: Parkinson’s disease is a major concern when the disease progresses to the middle stage of the illness. The typical features of Idiopathic Parkinson’s disease (IPD) are tremors impairment of the muscle tone, involuntary movement and bradykinesia. Improvement in strength and balance of IPD patients has improved their mobility functions. Especially, balancing exercises on uneven surfaces with eyes open and closed help them in gaining confidence to move outdoor independently with lesser risk of fall. Methodology: A total numbers of 30 subjects were considered for the study. All participants underwent two sets of measurement. Pre-test which was done at the beginning of the study & the post-test which was done at the end of 3 & 6 months of the study. 30 patients effectively completed the set of balancing and strengthening exercises with eyes closed & open for the period of 6 months. Results: All the subjects showed significant changes in BBS & ABC scales after 6 months of strength and balance training programs. The mean value of the pre- test scores were BBS – 37.23 ± 10.7 ABC – 57 ± 13.62 and post – test scores were BBS – 44.30 ± 8.78 ABC – 71.17 ± 13.62. Conclusion: From the statistical analysis it is evident that strengthening and balancing training program on uneven surfaces are effective in reducing the risk of fall and increasing the confidence of mobility in patients with PD. keywords: Parkinson’s disease; strengthening exercises; Therabands; balancing exercises. |
| Received on 15th August 2020, Revised on 27th August 2020, Accepted on 31st August 2020, DOI:10.36678/IJMAES.2020.V06I03.004 |
INTRODUCTION
Idiopathic Parkinson’s disease [IPD] is a group of the conditions affecting the motor system hence also called as motor system disorder. This is resulted due to the loss of dopamine producing in the brain cells.
Characteristics of Parkinson’s disease are progressive loss of muscle control, which leads to trembling of the limbs and head while at rest, stiffness, slowness, and impaired balance. As symptoms worsen, it may become difficult to walk, talk, and complete simple tasks1.
Most of the movement-related symptoms of IPD is considered as the second most common neurodegenerative disorders.2 When the amount of dopamine is too low, communication between the substantia nigra and corpus striatum becomes ineffective, and movement becomes impaired; the greater the loss of dopamine, the worse the movement-related symptoms. Other cells in the brain also degenerate to some degree and may contribute to non-movement related symptoms of Parkinson’s disease.3
The cause of Parkinson’s disease is unknown but researchers speculate that both genetic and environmental factors are involved; some genes have been linked to the disease. Although it is well known that lack of dopamine causes the motor symptoms of Parkinson’s disease, it is not clear why the Dopamine-producing brain cells deteriorate.
Genetic and pathological studies have revealed that various dysfunctional cellular processes, inflammation, and stress can all contribute to cell damage. In addition, abnormal clumps called Lewy bodies, which contain the protein alpha-synuclein, are found in many brain cells of individuals with Parkinson’s disease. The function of these clumps in regards to Parkinson’s disease is not understood. In general, scientists suspect that Dopamine loss is due to a combination of genetic and environmental factors2, 3.
Early symptoms of PD are subtle and occur gradually. In some people the disease progresses more quickly than in others.
The primary symptoms of Parkinson’s disease are all related to voluntary and involuntary motor function and usually start on one side of the body. Symptoms are mild at first and will progress over time. Some people are more affected than others are. Studies have shown that by the time that primary symptoms appear, individuals with Parkinson’s disease will have lost 60% to 80% or more of the Dopamine-producing cells in the brain. Characteristic motor symptoms include4:
- Tremors: Trembling in fingers, hands, arms, feet, legs, jaw, or head. Usually tremors occur while resting, but not while involved in a task. Tremors may worsen when a person is excited, tired, or stressed.
- Rigidity: Stiffness of the limbs and trunk, which may increase during movement. Rigidity may produce muscle aches and pain. Loss of fine hand movements can lead to cramped handwriting (micrographia) and may make eating difficult.
- Bradykinesia: Slowness of voluntary movement. Over time, it may become difficult to initiate movement and to complete movement. Bradykinesia together with stiffness can also affect the facial muscles and result in an expressionless, “mask-like” appearance.
- Postural instability: Impaired or lost reflexes can make it difficult to adjust posture to maintain balance. Postural instability may lead to falls.
- Parkinsonian gait: Individuals with more progressive Parkinson’s disease develop a distinctive shuffling walk with a stooped position and a diminished or absent arm swing. It may become difficult to start walking and to make turns. Individuals may freeze in mid-stride and appear to fall forward while walking.4
While the main symptoms of Parkinson’s disease are movement-related, progressive loss of muscle control and continued damage to the brain can lead to secondary symptoms. These secondary symptoms vary in severity, and not everyone with Parkinson’s will experience all of them, and may include. Anxiety, stress, confusion, memory loss or dementia, constipation, depression, difficulty in swallowing, excessive salivation, increased sweating, erectile dysfunction, skin problem, slowness of speech and monotone speech, incontinence of urinary or urgency for urination. 5
Several guidelines have been published to assist in the diagnosis of Parkinson’s disease. These include the Hoehn and Yahr scale and the Unified Parkinson’s Disease Rating Scale. Tests are used to measure mental capacity, behaviour, mood, daily living activities, and motor function. They can be very helpful in the initial diagnosis, to rule out other disorders, as well as in monitoring the progression of the disease to make therapeutic adjustments. Brain scans and other laboratory tests are also sometimes carried out, mostly to detect other disorders resembling Parkinson’s is disease.
The diagnosis of Parkinson’s disease is more likely if. At least two of the three major symptoms are present (tremor at rest, muscle rigidity, and slowness). The onset of symptoms started on one side of the body. Symptoms are not due to secondary causes such as medication or strokes in the area controlling movement. Symptoms are significantly improved with levodopa.
Researchers may disagree on the number of stages of Parkinson’s disease (range from 3-5 stages). However, they all agree the disease is a progressive disease with symptoms that usually occur in one stage may overlap or occur in another stage. The stage increase in number value for all stage naming systems reflects the increasing severity of the disease. The five stages used by the Parkinson’s Foundation are:
- Stage 1: mild symptoms (tremors and/or movement symptoms like swinging arm while walking) do not interfere with daily activities and occur on one side of the body.
- Stage 2: Symptoms worsen with walking problems and both sides of the body affected.
- Stage 3: Main symptoms worsen with loss of balance and slowness of movement.
- Stage 4: Severity of symptoms requires help; usually person cannot live alone.
- Stage 5: Caregiver needed for all activities; patient may not be able to stand or walk and may be bedridden and may also experience hallucinations and delusions.5,6
Parkinson’s disease cannot be cured completely but the symptoms can be relieved with the use of various medication with carbidopa is usually given for the PD treatment, Carbidopa helps in delay of conversion of levodopa into dopamine. The nerve cells use levodopa for the production of dopamine and thus replenish the supply deficiency of the brain’s dopamine7.
Thus, levodopa is very helpful (at least ¾) of Parkinson’s cases. Not all Parkinson’s symptoms respond equally to this drug. Tremors don’t have much effect but bradykinesia and rigidity is remarkably reduced. Balance issues and other symptoms may not be alleviated at all. Anti-cholinergic have a great effect in controlling tremors and rigidity. Bromocriotine, Pramipexole and ropinirole, mimics the role of dopamine thus helping the neurons to use it as dopamine.8
An antiviral drug amantadine also helps in reducing the symptoms. In May 2006, FDA also approved the drug called Safinamide, which can be used for diminishing the experience of “off” periods or patients with increasing symptoms of PD. In some cases, surgery can also be done for the patients not responding well to drugs. Deep brain stimulation (DBS) is now approved by U.S FDA (Food and Drug administration) where an electrode is implanted in the brain and is connected to an electrical device called pulse generator which can be externally programmed.
This process of stimulation reduces the need of drugs thus decreasing the involuntary movements call dyskinesia which is a common side effect of these drugs. This procedure of stimulation to brain also reduces tremors, slowness and gait disturbances. DBS requires careful programming in order to work correctly3, 8, 9.
Fall is very common in PWP. Gait impairment, freezing of gait, cognition, loss of postural control is the common cause of falling. This is not easily managed by medications only
Frequent falls can cause loss of mobility, restriction in daily living activities, fractures and cost of treatment is increased 10, 11.
Studies have shown that exercises can be useful in preventing falls in PwP (patients with Parkinson’s disease).
Physiotherapy along with drug therapy is the most commonly used procedure for PwP. However, the Cochrane reviews have supported this procedure with many randomized control trials 7, 8, 12.
Many authors have suggested that balance impairment in PD and normal old age changes causes decrease in the muscle strength due to their sedentary lifestyle. It has been noted that strengthening and balancing rehabilitation programs have reduced the risk of falls, prevent dysfunction and dependency in the elderly 13, 20, 21.
METHODOLOGY
Study design: An Interventional Study
Study population: Subjects who are diagnosed with IPD by their Neurologist
Study setting: The study was conducted (testing & Intervention) at Outpatient department of Bethel Medical Mission HOSPITAL.
Study sample size A total of 30 patients
Sampling Method: Purposive sampling
Study duration: 6 months
Selection criteria
Inclusion criteria:
- Diagnosed with IPD by their neurologist
- Ambulatory and able to follow simple commands
- Patient with Unified Parkinson’s Disease Rating scale score of 35 and above
- Patient with a score of above 40% on the Activities Specific Balance Confidence Scale (ABC).
Exclusion criteria:
- Suffering from unstable cardiovascular disease
- Uncontrolled chronic conditions that might interfere with the safety and conduct of the training and testing protocol.
- Patients participated earlier in balance and strengthening program
Outcome measures tools used for the study
- The Berg Balance Scale (BBS)
- The Activities-Specific Balance Confidence Scale (ABC)
Material used: Data collection Sheets, Stop watch,15 ft Walk way, 4-inch Foam Pads, Thera bands, Chairs, Weight cuffs and Thera tubes.
Methods: A total of 30 subjects fulfilling the selection criteria were included in the study after taking informed consent from each one of them. The Unified Parkinson’s Disease Rating Scale (UPDRS) Score is used for their eligibility.
The Unified Parkinson’s disease rating scale (UPDRS) has 4 sections
I-Mentation behaviour and mood
II- ADL activities
III- Motor examination
IV- Complication of therapy
Score – 0 to 147
Higher the score = Worst performance 12,15.
Baseline evaluation of Balance was be done using The Berg Balance Scale (BBS) Score, The Activities-Specific Balance Confidence Scale (ABC).
All participants received the same Balancing intervention and Muscle strength intervention for 6 months. Outcome measurements of Balance and Muscle strength were assessed using The Berg Balance Scale (BBS), The Activities-Specific Balance Confidence Scale (ABC) at the end of 3 months & 6 months13,17.
Use of outcomes measures tools
All patients were evaluated at baseline and at the end of 3 months & 6 months of treatment period by the same examiner using Berg Balance Scale (BBS) 14 items (0-4 points per task higher score=best performance).
This scale evaluates balance during activities like sitting, standing and positional changes.
The Activities specific balance confidence scale (ABC) is the scale which examines patients perceived level of balance confidence while doing 16 activity of daily living rated from 0 to 100 each14, 16.
Procedure for intervention
Balancing exercise s were given thrice a week and strengthening exercises were given on remaining 3 days a week.
Thus, the duration for balance training was 30 minutes and strength training was for 15 minutes. Frequency of training – 3 days a week each for 6 days.
Balance Intervention:
Balance exercise session lasted for 30 min and was conducted on 3 non- consecutive days, every week. Balance training programme include standard rehabilitation exercises for balancing. This training improved balance in older adults with PD15, 16, 20.
Training was in 2 parts :
1.Standing on a 4-6-inch-thick foam pad with feet- shoulder width apart with eyes open and then eyes closed along with neck in neutral followed by neck extension for 20 sec. Repeated for 5 times.
2.Standing with feet – shoulder width apart without the foam pad with eyes open and then eyes closed along with neck in neutral followed by neck extension for 20 sec. Repeated for 5 times.
Muscle strengthening intervention:
Strengthening exercises were done with weight cuffs, TheraBand & Theratubes. All participants had undergone progressive strengthening of trunk, hip, knee, and ankle.
The training protocol used standard principle of rehabilitation of using concentric and eccentric muscles strength.
RESULTS
Primary analysis:
Pairwise comparisons were done for scores of BBS and ABCS using paired t-test.
Secondary analysis:
Correlation between Age and UPRSD was done using Karl Pearson’s correlation coefficient.
Comparison of change scores and UPRSD between gender was done using Independent t-test.
All analyses were done at 95% confidence interval using Statistical package for social sciences (SPSS version 22, Chicago, IL) for Windows software.
Tables and Graphs:
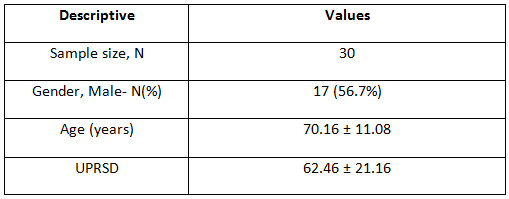
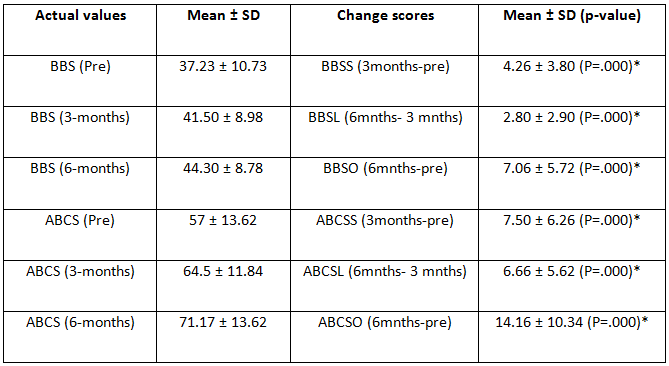
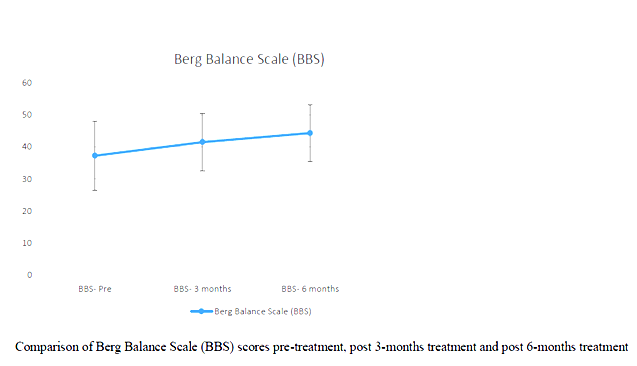
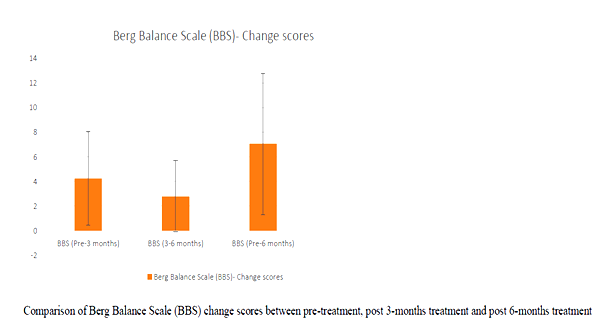
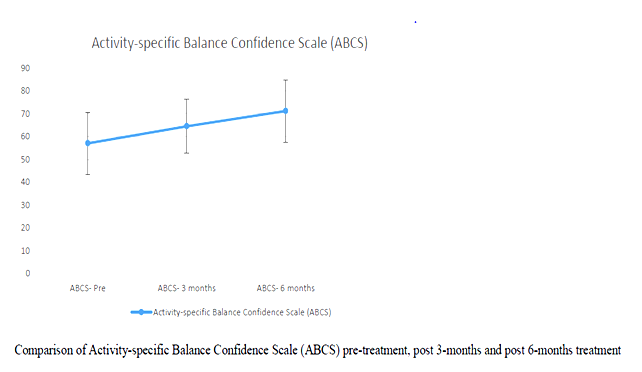
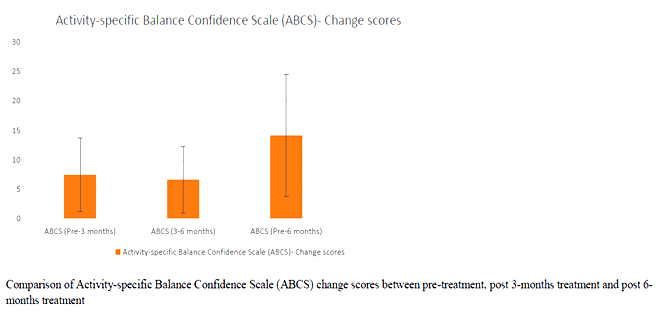
Correlation between age and UPRSD: Weak negative correlation existed (-.123) which was not statistically significant (p=.518)
Comparison of UPRSD between Genders:

Comparison of BBS and ABCS change scores between Genders:
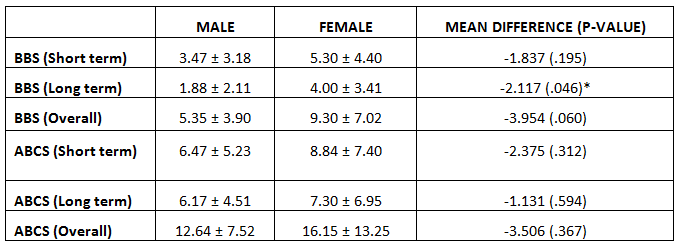
BBS change score between 3-months and 6-months was better amongst women than men, statistically significant at p=.046. All other change scores were not influenced by gender (p>.05).
DISCUSSION
The main aim of the study was to evaluate disease specific and balance related measure in the given population. The clinical scales used in the study are sensitive in the evaluation of the risk of fall in patients with Idiopathic Parkinson’s Disease. Cut-off scores of these scales are very useful in clinical practice as it provides detailed description about impaired functional activities and balance related activities & can also be used to evaluate treatment outcomes.
The present study shows that balance and strengthening exercises together help in reducing risk of fall and improves functional mobility in patients with Idiopathic Parkinson’s disease.
In the study BBS and ABC pre and post test scores were analysed. It has been noted than all of the participants improved in their BBS and ABC scores.
BBS change score between 3-months and 6-months was better amongst women than men, & statistically significant at p=.046. All other change scores were not influenced by gender (p>.05). Similar study was done in the year 2003 & 2015 where they studied the Effect of balance and resistance training using computerized dynamic posturography (sensory orientation test) SOT for balance and muscle strength in 15 patients with IPD. Authors concluded that balance and strength of muscles can be improved in patient with PD by training programmes of balance and high intensity resistance 12, 19.
A systematic review study reported the evidence of resistance training on the strength and function in patients with PD. The study demonstrated that moderate intensity training for 2-3 times per week over 8-10 weeks can result in significant improvement in strength, balance and others motor symptoms in patients with early to moderate stage of PD18,20,21.
Another randomized control trial of 210 patients with PD were divided into 3 groups and were educated for balance training movement strategy training and strength training programs. The study concluded that rehabilitation training reduces the risk of falls in patients of mild to moderate stage of PD 19, 20
The present study results provide validation and best combination of outcome measures used in PD. With these scales risk of recurrent falls too can be determined. Although the patients performed well according to the scales, but an independent validation of sample is important in order to use into clinical practice.
Ethical clearance: The ethical approval was granted by the ethical committees of the Hosmat College of Physiotherapy, Bangalore.
Conflict of interest: There was no conflict of interest to conduct this study.
Fund for the study: It was self-financed study.
Limitations: Small sample size, smaller time period of study, Frequent follow up.
Recommendations: Early stages of Parkinson’s and larger study size
CONCLUSION
The study result showed that Strengthening and balancing exercises have a great effect on patients with Parkinson’s disease. It delays the progress of the Disease and helps the patients to regain their confidence in mobility and become more active and independent. According to the statistical analysis female patients showed better response to the training as compared to the male patients.
REFERENCES
- Parkinson’s disease information page (January 18, 2019). National Institutes of Neurological Disorders and Stroke website. ninds.nih.gov/disorders/All-Disor ders/ Parkinsons Disease Information-Page. Updated June 12, 2108.
- Kalia L V. et al.(August 2015). Parkinson’s disease; 386(9996); 8; 96-912. PMID 25904081.
- Sveinbjornsdottir. S (Oct 2016). Quit;”The clinical symptoms of Parkinson’s disease” ; Journal of Neurochemistry.139 Suppl 1; 318-324; Bibcode: 2006JNeur. 26.9606G.
- Braak, H et al. (1998). Staging of brain pathology related to patients with Parkinson’s disease. C. An algorithm (decision tree) for the management of Parkinson’s disease. Neurology; 50 Suppl 3; S157.
- Poewe W (December 2006). “The natural history of Parkinson’s disease”. Journal of Neurology. 253 Suppl 7 (Suppl 7); VII2–6.
- Marchese R, Bove M, Abbruzzese G. (2003).Effect of cognitive and motor tasks on postural stability in Parkinson’s disease: a posturographic study. Mov Disord., 18; 652 658.
- Deane KHO, Jones D, Ellis-Hill C, Clarke CE, Playford ED, Ben-Shlomo Y. (2001). Physiotherapy for Parkinson’s disease: A comparison of techniques. Cochrane Database Syst Rev. : CD002815.
- Deane KHO, Jones D, Playford ED, Ben-Shlomo Y, Clarke CE. (2001). (Physiotherapy versus placebo or no intervention in Parkinson’s disease. Cochrane Database Syst Rev. 3):CD002817.
- Olanow CW, Koller WC. (1998). An algorithm (decision tree) for the management of Parkinson’s disease. Neurology, 50 Suppl 3; S157.
- Chung CL, Thilarajah S2, Tan D3. (2016 Jan). Effectiveness of resistance training on muscle strength and physical function in people with Parkinson’s disease: a systematic review and meta-analysis. Clin. Rehabil., 30(1); 11-23.
- Morris ME, Menz HB, McGinley JL, Watts JJ, Huxham FE, Murphy AT, Danoudis ME, Iansek R. (2015 Sep). A Randomized Controlled Trial to Reduce Falls in People With Parkinson’s Disease.Neurorehabil Neural Repair. 29 (8); 777-85.
- Conradsson D, Löfgren N, Nero H, Hagströmer M, Ståhle A, Lökk , Franzén E. (2015 Oct). The Effects of Highly Challenging Balance Training in Elderly With Parkinson’s Disease: A Randomized Controlled Trial. Neurorehabil Neural Repair., 29(9); 827-36.
- Corcos DM, Robichaud JA, David FJ, Leurgans SE, Vaillancourt DE, Poon C, Rafferty MR, Kohrt WM, Comella CL.(2013 Aug). A two-year randomized controlled trial of progressive resistance exercise for Parkinson’s disease. Mov Disord. 28(9); 1230-40.
- Olanow CW, Wunderle KB, Kieburtz K. (2011 May). Milestones in movement disorders clinical trials: advances and landmark studies.MovDisord.,26(6); 1003-14.
- Glendinning DS1, Enoka RM. (1994 Jan). Motor unit behavior in Parkinson’s disease. Phys Ther., 74(1); 61-70.
- Smania N, Corato E, Tinazzi M, Stanzani C, Fiaschi A, Girardi P, Gandolfi M. (2010 Nov-Dec). Effect of balance training on postural instability in patients with idiopathic Parkinson’s disease. Neuro-rehabil Neural Repair.,24(9); 826-34.
- Qutubuddin AA, Pegg PO, Cifu DX, Brown R, McNamee S, Carne W. (2005 Apr). Validating the Berg Balance Scale for patients with Parkinson’s disease: a key to rehabilitation evaluation.Arch Phys Med Rehabil., 86(4); 789-92.
- Hirsch MA, Toole T, Maitland CG, Rider RA. (2003 Aug). The effects of balance training and high-intensity resistance training on persons with idiopathic Parkinson’s disease. Arch Phys Med Rehabil., 84(8); 1109-17.
- Inkster LM1, Eng JJ, MacIntyre DL, Stoessl AJ. (2003 Feb). Leg muscle strength is reduced in Parkinson’s disease and relates to the ability to rise from a chair. MovDisord. 18(2); 157-62.
- Scandalis , T , et al. (2001).Resistance Training and Gait function in patients with Parkinson’s disease. Am J Phys Med Rehabil. 80; 38.
- Glendinning, D: (2001). A rationale for strength training in patients with Parkinson’s disease. Neurology report (now JNPT); 21; 132.
| Citation: Purnima Singh, Panomootil Blessy Varghese (2020).effects of balance training and strengthening exercises on individuals with idiopathic parkinson’s disease, ijmaes; 6 (3); 799-810. |


Leave a Reply